
One of the most often utilised tissue patches following an arteriotomy during cardiovascular surgery is the bovine pericardium patch, which is commonly used in surgery. You can purchase items from Bridge Med Solutions for a fair price. Call us right away for further details. Contact us with any questions! boardboardboardboardboard

BridgeMed Solutions is the perfect extension of your in-house R&D department. Our Heart Valve Startup Incubators are manufactured in our state-of-the-art facility. We provide Heart Valve Venture Capital and solutions for your device. Call us now! board
We provide Bovine Pericardium Tissue in the USA. We are the first to cross-link bovine and porcine tissue to meet the exact specifications of transcatheter heart valves. Contact us today for any inquiry! board
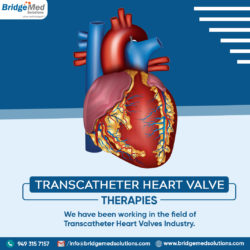
Whether you are start-ups or large international device firms, we are ideally positioned to advise and help businesses in the TAVR, TMVR, and Pulmonary/Tricuspid space flourish. Contact us now!

For manufacturers’ needs, Bridgemed Solutions offers a range of bovine pericardium tissue harvesting and selection alternatives. For more information on our Bovine Pericardium Tissue Patch options, please get in touch with us. boardboardboardboardboard

One of the most often used patches following an arteriotomy during cardiovascular surgery is a bovine pericardium tissue patch, which is widely used in surgery. This patch is available from Bridge Med Solutions at a reasonable price. Contact us for any questions or concerns.
Both pericardial tissue and Elgiloy stent material are known for having the best intrinsic qualities for valve manufacturing. It combines the benefits of bovine pericardial tissue with the latest engineering advances. Contact us now for any inquiry! TISSUE PATCH FOR SALE
We are a prominent tissue patch maker for heart valves. Bridgemed Solutions, as a leader in heart valve therapy, remains committed to providing cardiac surgeons with dependable solutions from a firm they can trust. Contact us now! boardboard

We are a group of highly competent PhD scientists, tissue engineers, delivery system and stent experts, and skilled technicians who will blend in seamlessly with your R&D and operations teams. Our employees collaborate with your firm at various levels to find and offer ways that save time and money. Contact us now!

BridgeMed Solutions is one of the best firms for contract manufacture of TAVR and TAVI under the supervision of professionals. The doctor will select whether the TAVR or TAVI method will be used for your heart surgery based on your condition. Contact us now! boardboardboardboard
The bovine pericardium tissue patch is widely used in surgery and is one of the most common patches used after an arteriotomy during cardiovascular surgery. This patch is available from Bridge Med Solutions at a reasonable price. Contact us now! boardboardboardboardboardboardboardboard
BridgeMed Solutions offers complete contract manufacturing services for TAVR, TMVR, Tricuspid, and Pulmonic Valves. Get in touch with us now for any inquiry.

Bridgemed provides a variety of tissue heart valves to help patients with valvular heart disease. Our aortic and mitral tissue valves are made from specially selected and treated bovine or porcine tissue that closely resembles human heart valves. Contact us now!

BridgeMed Solutions produces custom transcatheter aortic valve (TAVR), transcatheter mitral valve (TMVR), and transcatheter tricuspid valves from scratch per client requirements. Get in touch with us now for any inquiry. boardboardboardboardboardboard

The bovine pericardium tissue patch is widely used in surgery and is one of the most common patches used after an arteriotomy during cardiovascular surgery. From Bridge Med Solutions you can buy at reasonable prices. Contact us now for more information.
Bridgemed provides a variety of tissue heart valves to help patients with valvular heart disease. Our aortic and mitral tissue valves are made from specially selected and treated bovine or porcine tissue that closely resembles human heart valves. Call us now for any inquiry.

Bridge Med Solutions provides high-quality bovine pericardial tissue, which is an excellent choice for all cardiac and vascular treatments. Cardiac surgeons make great use of it. Get in touch with us for any help.

It’s critical to maintain good health in order to improve cardiac function. Staying on top of your heart valve and blockage treatment is critical. At a modest cost, we provide Heart Valve Testing – AWT, Durability, and Hydrodynamic. Contact us now!

We collaborate with physicians to develop solutions that allow people to live longer, healthier, and more productive lives. We are particularly interested in medical breakthroughs that address vast and expanding patient populations with unmet clinical needs. Get in touch with us now for any enquiry.
Transcatheter Valve Consulting is a cutting-edge approach to medical devices designed for cardiac therapy. It entails cross-linking, tissue cleansing, stent delivery, and setting up production facilities for tissue fixation, among other things. Consulting Services boardboardboardboardboard


